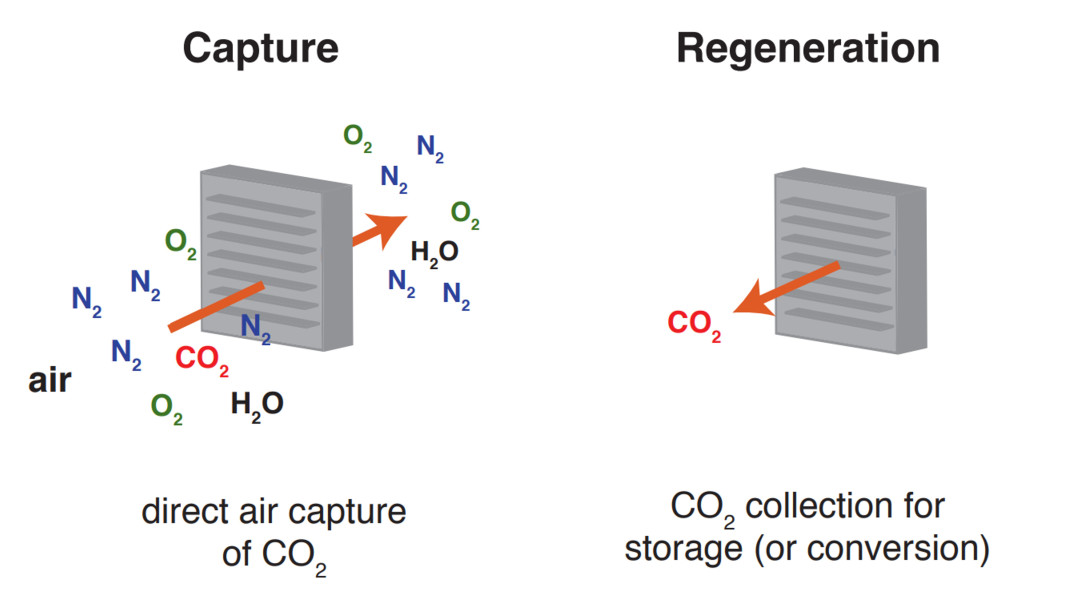
Direct air capture of carbon dioxide
The urgent need to reverse the rising concentration of carbon dioxide (CO2) in the atmosphere has created a strong demand for affordable and scalable carbon capture technologies. Direct air capture (DAC), which removes CO2 directly from ambient air, has gained significant attention as a key strategy for addressing emissions that have already been released. However, most current DAC technologies, based on solid amines or alkaline solutions, remain expensive and energy intensive. Researchers are developing low-cost sorbents that could help address the financial and energy costs.

Sheet of sorbent material for DAC, developed by the Forse lab.
The idea
Activated charcoal is a low-cost sorbent widely used in industrial applications such as water filtration and carbon capture. However, it cannot effectively capture CO2 from air because of the low atmospheric CO2 concentration. A promising solution is to load chemical compounds that can form reversible bonds with CO2 into the pores of activated charcoal, forming low-cost DAC sorbents.
Using this sorbent, CO2 capture operates in a continuous cycle, with adsorption at low temperature and regeneration at higher temperature to release the captured CO2. During regeneration, because activated charcoal is electrically conductive, Joule heating offers a simple and efficient way to supply heat by passing electricity directly through the sorbent. When powered by renewable electricity, such as solar or wind, we can drive carbon capture from air in a cleaner and more sustainable way.
This DAC project is being developed by a multidisciplinary team from the Department of Chemistry and the Department of Engineering, led by Prof. Alex Forse and Prof. Stuart Scott.






